 International Journal of Antimicrobial Agents. Dec;56(6):106214.
International Journal of Antimicrobial Agents. Dec;56(6):106214.
Most studies of Covid-19 involve very sick hospitalized patients. The authors are testing the use of Hydroxycholoroquine (HCQ) plus zinc and an antibiotic (for prevention of secondary lung infection) as an early treatment beginning within the first week of illness.
Dr. Zelenko, the medical doctor part of this group, treated 141 patients, ages 40-67 years and with confirmed Covid-19 disease. The patients were chosen from a group of consecutive adult patients diagnosed with Covid-19 and who were also either over 60 years old or had high blood pressure, high cholesterol, diabetes, obesity, heart, kidney or liver disease, history of blood clots, or asthma, lung disease, autoimmune disease, or cancer. One pregnant woman was included.
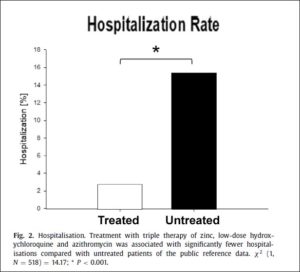
712 patients in the community were diagnosed with Covid-19 by PCR testing, and 37 more were confirmed by IgG testing. Of those meeting the risk criteria, 377 patients received “standard care” by other practitioners in the community and were the Control Group, while 141 were treated by Dr. Zelenko’s protocol as the Intervention (Treated) Group. Outcomes were as follows:
- HOSPITALIZED:
- Treated Group: 2.8%
- Control Group: 15.4%
- Treated Group: 2.8%
- DIED:
- Treated Group: 0.71% (1 person)
- Control Group: 3.5 % (13 people)
The Zelenko Protocol
Dr. Zelenko himself feels his protocol can save many lives, so he has provided it for free to all who want it: Treatment Protocol || Prophylaxis Protocol
 Or download from the links below:
Or download from the links below:
Treatment_Protocol || Prophylaxis-Protocol
He has also provided the following graphic to illustrate how it works (click to enlarge):























